Class 10 | science
Chemical Reactions and Equations
Learn about chemical reactions, balancing equations, types of reactions, corrosion and rancidity.
Theory
Learn about chemical reactions, balancing equations, types of reactions, corrosion and rancidity.
Concept 1
Introduction to Chemical Reactions
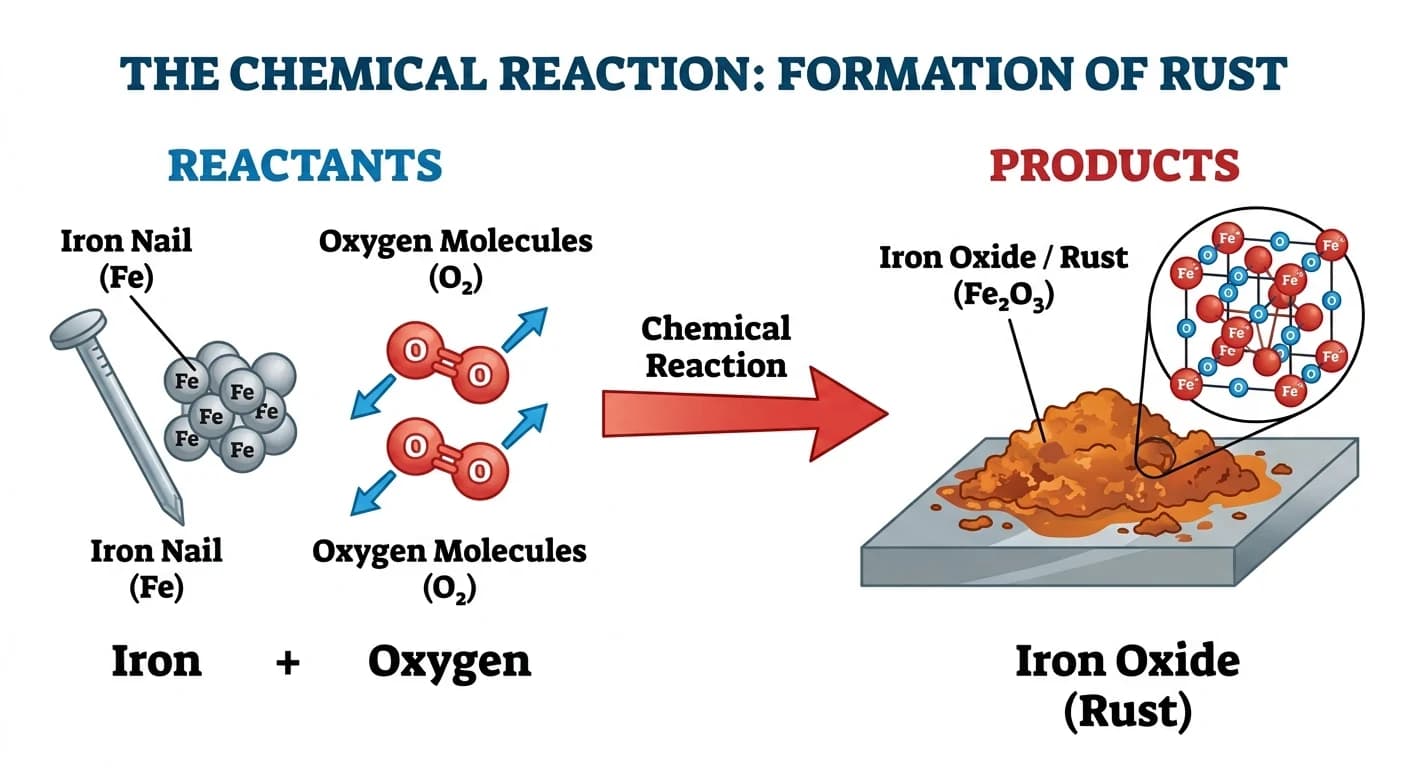
In a chemical reaction, the atoms of reactants rearrange to form new substances. The original substances are called reactants and the new substances formed are called products. For example, when iron rusts, iron reacts with oxygen to form iron oxide — a completely new substance.
A chemical reaction is a process where one or more substances (reactants) are transformed into one or more new substances (products) with different properties.
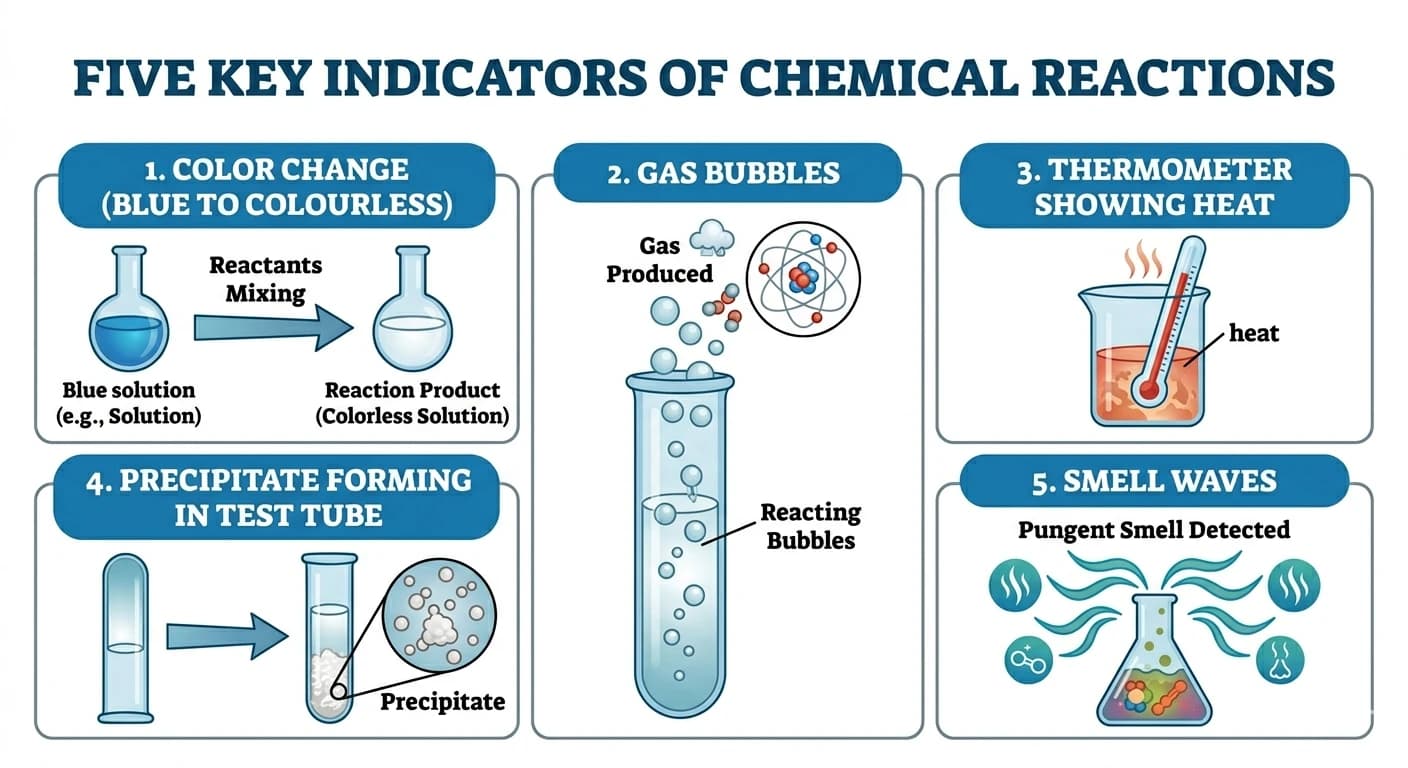
These signs tell us something new has been formed. For example, when we burn magnesium, it gives a bright white light and forms white ash — colour change and energy change both happen. When vinegar and baking soda mix, bubbles of CO₂ gas are formed — that's a gas evolution sign.
Signs of a chemical reaction include
- (1)Change in colour,
- (2)Evolution of a gas (bubbles),
- (3)Change in temperature (heat given out or absorbed),
- (4)Formation of a precipitate (solid formed in a liquid),
- (5)Change in smell.
Concept 2
Chemical Equations
A word equation describes a reaction in words (e.g., Magnesium + Oxygen → Magnesium Oxide). A chemical equation replaces words with chemical symbols and formulas, making it shorter and universal. The arrow (→) means 'reacts to form' or 'yields'.
A chemical equation is a symbolic representation of a chemical reaction using chemical formulas. Reactants are written on the left side and products on the right side, separated by an arrow (→).
Example: Mg + O₂ → MgO
Antoine Lavoisier proposed the Law of Conservation of Mass. This means the total mass of reactants = total mass of products. If we don't balance an equation, it would incorrectly show atoms appearing or disappearing, which is not possible. Example: H₂ + O₂ → H₂O is unbalanced. Balanced: 2H₂ + O₂ → 2H₂O
A balanced chemical equation has equal number of atoms of each element on both the reactant side and the product side.
We balance equations because of the Law of Conservation of Mass — matter cannot be created or destroyed in a chemical reaction.
Writing physical states makes the equation more informative. For example, (aq) means the substance is dissolved in water. The upward arrow ↑ after a gas formula shows that the gas is released (evolved). The downward arrow ↓ after a formula shows that a solid precipitate is formed.
These symbols show the physical state of substances:
(s) = solid
(l) = liquid
(g) = gas
(aq) = aqueous solution (dissolved in water)
Example: Zn(s) + H₂SO₄(aq) → ZnSO₄(aq) + H₂(g)↑
Concept 3
Types of Reactions — Combination
The word 'combination' means 'joining together'. In these reactions, simpler substances join to make a more complex one. Burning of coal (C + O₂ → CO₂) and formation of water from hydrogen and oxygen (2H₂ + O₂ → 2H₂O) are also examples. Burning of coal in air is exothermic — it releases heat.
- (1)2Mg + O₂ → 2MgO (Magnesium burns in oxygen)
- (2)CaO + H₂O → Ca(OH)₂ (Quicklime reacts with water)
Concept 4
Types of Reactions — Decomposition
- (1)Thermal: 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂ (lead nitrate heated)
- (2)Electrolytic: 2H₂O → 2H₂ + O₂ (water split by electricity)
- (3)Photolytic: 2AgCl → 2Ag + Cl₂ (silver chloride in sunlight turns grey)
- (1)Thermal decomposition — by heat
- (2)Electrolytic decomposition — by electricity
- (3)Photolytic decomposition — by light
Concept 5
Types of Reactions — Displacement
The reactivity series of metals helps us predict which metal will displace which. More reactive metal always displaces less reactive metal. When zinc is placed in copper sulphate solution, the blue colour of CuSO₄ fades as colourless ZnSO₄ forms and reddish-brown copper deposits on zinc.
- (1)Zn + CuSO₄ → ZnSO₄ + Cu (Zinc displaces copper)
- (2)Fe + CuSO₄ → FeSO₄ + Cu (Iron displaces copper)
- (3)Zn + 2HCl → ZnCl₂ + H₂↑ (Zinc displaces hydrogen from acid)
Concept 6
Types of Reactions — Double Displacement
The word 'double' tells us that both compounds exchange partners. If a precipitate (insoluble solid) is formed, it is also called a precipitation reaction. Another example: AgNO₃ + NaCl → AgCl↓ + NaNO₃ — white precipitate of silver chloride forms. These reactions are used in water treatment and in qualitative analysis.
In a double displacement reaction, two compounds exchange their ions to form two new compounds. It is also called a metathesis reaction.
General form: AB + CD → AD + CB
Example: Na₂SO₄ + BaCl₂ → BaSO₄↓ + 2NaCl
Here, barium sulphate (BaSO₄) forms as a white precipitate.
Concept 7
Oxidation and Reduction
A simple way to remember: OIL RIG — Oxidation Is Loss (of electrons), Reduction Is Gain (of electrons). In the example, copper oxide (CuO) gives its oxygen to hydrogen. So CuO is reduced (loses oxygen) and H₂ is oxidised (gains oxygen). Both oxidation and reduction happen together — you cannot have one without the other!
- CuO loses oxygen → CuO is reduced
- H₂ gains oxygen → H₂ is oxidised
Redox = Reduction + Oxidation happening at the same time. You cannot have oxidation without reduction. Think of it as a transfer: one substance loses electrons/oxygen while the other gains them. In the above example, oxygen is transferred from CuO to H₂. The reducing agent reduces others but gets oxidised itself.
- H₂ is oxidised → H₂ is the reducing agent
- CuO is reduced → CuO is the oxidising agent
Concept 8
Effects of Oxidation — Corrosion
- Damages machinery, vehicles, bridges, iron railings
- Weakens metal structures
- Huge economic loss every year
- Painting or greasing the surface
- Galvanisation (coating iron with zinc)
- Alloying (e.g., stainless steel has chromium added to iron)
- Electroplating with a non-reactive metal
- (1)Rusting of iron: 4Fe + 3O₂ + xH₂O → 2Fe₂O₃·xH₂O (rust)
- (2)Black coating on silver: 2Ag + H₂S → Ag₂S + H₂
- (3)Green coating on copper: Copper reacts with CO₂ and moisture → basic copper carbonate (green)
Concept 9
Effects of Oxidation — Rancidity
Rancidity is a type of slow oxidation. The fats in butter, ghee, chips, biscuits can become rancid over time. You may have noticed chips packets being filled with nitrogen gas — this prevents the chips from going stale. Antioxidants like vitamin C (ascorbic acid) and vitamin E are also natural antioxidants added to some foods. Rancidity changes the smell, taste, and nutritional value of food.
- (1)Adding antioxidants (like BHA, BHT) to food
- (2)Storing food in airtight containers (reduces oxygen contact)
- (3)Refrigeration (slows down oxidation)
- (4)Flushing packets with nitrogen gas (inert — doesn't react with food)
- (5)Vacuum packing
Concept 10
Exothermic and Endothermic Reactions
Easy way to remember: 'Exo' means outside — heat goes OUT (released). 'Endo' means inside — heat goes IN (absorbed). Everyday example: Burning LPG in a gas stove releases heat — exothermic. Photosynthesis absorbs sunlight energy — endothermic. When we dissolve ammonium chloride in water, the test tube feels cold — endothermic reaction.
Exothermic reaction: A reaction that releases energy (heat) to the surroundings. The temperature of surroundings increases.
Examples: Burning of coal, respiration, neutralisation of acid and base
Equation: C + O₂ → CO₂ + heat energy
Endothermic reaction: A reaction that absorbs energy (heat) from the surroundings. The temperature of surroundings decreases.
Examples: Decomposition of limestone, photosynthesis, dissolving ammonium chloride in water
Equation: CaCO₃ + heat → CaO + CO₂
Key Points
- A chemical reaction converts reactants into products with new properties.
- A chemical reaction is indicated by colour change, gas evolution, temperature change, precipitate formation, or smell change.
- Chemical equation = reactants → products, written using chemical formulas.
- Balanced equation follows Law of Conservation of Mass — atoms are neither created nor destroyed.
- (s)=solid, (l)=liquid, (g)=gas, (aq)=dissolved in water. ↑=gas evolved, ↓=precipitate formed.
- Combination reaction: Two or more substances combine → one new product. A + B → AB
Questions
Quick Check
Which of the following is NOT a sign of a chemical reaction?
Show AnswerHide Answer
(B) Change in shape
Explanation
Change in shape is a physical change, not a chemical change. For example, cutting a paper into pieces changes its shape but no new substance is formed. Chemical reactions always involve formation of new substances, which is shown by colour change, gas evolution, temperature change, precipitate formation, or change in smell.
Quick Check
When magnesium burns in air, the product formed is
Show AnswerHide Answer
(B) Magnesium oxide
Explanation
When magnesium (Mg) burns in oxygen (O₂) from air, it undergoes a combination reaction to form magnesium oxide (MgO). The reaction is: 2Mg + O₂ → 2MgO. A bright white flame is seen during this reaction, which is a sign of a chemical reaction taking place.
Quick Check
The chemical equation: 2H₂ + O₂ → 2H₂O is
Show AnswerHide Answer
(B) A balanced equation
Explanation
In 2H₂ + O₂ → 2H₂O: Left side has 4 H atoms (2×2) and 2 O atoms. Right side has 4 H atoms (2×2) and 2 O atoms (2×1). Both sides are equal, so the equation is balanced. It correctly follows the Law of Conservation of Mass.
In-content Ad
AdSense placeholder
Quick Check
Balance the following chemical equation: Fe + H₂O → Fe₃O₄ + H₂
Show AnswerHide Answer
3Fe + 4H₂O → Fe₃O₄ + 4H₂
Explanation
- Right side has 3 Fe atoms in Fe₃O₄, so put 3 before Fe on left: 3Fe
- Right side has 4 O atoms in Fe₃O₄, so we need 4 H₂O on left: 4H₂O
- 4H₂O gives 8 H atoms on left, so we need 4H₂ on right
- Check: Fe: 3=3 ✓, O: 4=4 ✓, H: 8=8 ✓
Quick Check
When quicklime (CaO) is added to water, the reaction that occurs is
Show AnswerHide Answer
(B) Combination reaction
Explanation
CaO + H₂O → Ca(OH)₂. Here, quicklime (CaO) and water (H₂O) combine to form a single product — calcium hydroxide Ca(OH)₂ (also called slaked lime). Since two substances are joining to form one, it is a combination reaction. This reaction is also highly exothermic — a lot of heat is released.
Quick Check
The decomposition of silver chloride in presence of sunlight is an example of
Show AnswerHide Answer
(C) Photolytic decomposition
Explanation
2AgCl(s) → 2Ag(s) + Cl₂(g). This reaction occurs in presence of sunlight (light energy). Since light causes the decomposition, it is called photolytic decomposition. The white silver chloride turns grey because silver metal is formed. This principle is used in black and white photography.
In-content Ad
AdSense placeholder
Quick Check
When ferrous sulphate crystals are heated, what is observed?
Show AnswerHide Answer
(C) Crystals turn reddish-brown and SO₂/SO₃ gas is released
Explanation
FeSO₄·7H₂O (ferrous sulphate, green crystals) when heated: FeSO₄(s) → Fe₂O₃(s) + SO₂(g) + SO₃(g). The green crystals lose water first and turn white, then decompose to form reddish-brown iron oxide (Fe₂O₃), and release SO₂ and SO₃ gases with a suffocating smell. This is a thermal decomposition reaction.
Quick Check
When iron nails are kept in copper sulphate solution, the colour of solution changes from
Show AnswerHide Answer
(A) Blue to green
Explanation
Fe + CuSO₄ → FeSO₄ + Cu. Iron (Fe) displaces copper from copper sulphate solution. The blue colour of copper sulphate (CuSO₄) fades because it is consumed, and green iron sulphate (FeSO₄) is formed. Reddish-brown copper deposits on the iron nail. Iron is more reactive than copper, so it displaces copper.
Quick Check
In the reaction: Na₂SO₄ + BaCl₂ → BaSO₄↓ + 2NaCl, what type of reaction is it?
Show AnswerHide Answer
(C) Double displacement reaction
Explanation
In this reaction, Na₂SO₄ and BaCl₂ exchange their ions — sodium (Na⁺) swaps with barium (Ba²⁺). Two new compounds are formed: BaSO₄ (barium sulphate — a white precipitate, shown by ↓) and NaCl (sodium chloride). Since both compounds exchange ions, it is a double displacement reaction. The ↓ symbol shows BaSO₄ is an insoluble precipitate.
In-content Ad
AdSense placeholder
Quick Check
Identify the type of reaction for each
Show AnswerHide Answer
Explanation
Quick Check
In the reaction: ZnO + C → Zn + CO₂, which substance is oxidised?
Show AnswerHide Answer
(C) C (Carbon)
Explanation
- Carbon (C) gains oxygen (from ZnO) to form CO₂ → Carbon is oxidised (gains oxygen)
- ZnO loses oxygen to form Zn → ZnO is reduced (loses oxygen)
Quick Check
In the reaction: MnO₂ + 4HCl → MnCl₂ + 2H₂O + Cl₂, HCl is
Show AnswerHide Answer
(C) Both oxidised and reduced
Explanation
- (1)Some HCl molecules lose hydrogen (oxidised) — their H joins O to form H₂O
- (2)Some HCl molecules lose Cl⁻ which forms Cl₂ gas — the Cl is oxidised
In-content Ad
AdSense placeholder
Quick Check
Galvanisation is a process used to prevent rusting of iron. In this process, iron is coated with
Show AnswerHide Answer
(C) Zinc
Explanation
Galvanisation is the process of coating iron or steel with a thin layer of zinc to protect it from rusting. Zinc is more reactive than iron, so it reacts with oxygen and moisture instead of iron, protecting the iron beneath. Zinc forms a protective layer of zinc oxide/carbonate on its surface. Galvanised iron pipes and buckets are common examples.
Quick Check
The green coating seen on old copper vessels is due to
Show AnswerHide Answer
(B) Copper reacting with CO₂ and moisture to form basic copper carbonate
Explanation
When copper is exposed to air (which contains CO₂ and moisture), it slowly forms a green layer of basic copper carbonate: Cu + CO₂ + H₂O → CuCO₃·Cu(OH)₂ (basic copper carbonate). This is why old copper statues, vessels, and the Statue of Liberty appear green. This is a form of corrosion specific to copper.
Quick Check
Why do we apply paint or oil to iron objects? Explain with a chemical reason.
Show AnswerHide Answer
We apply paint or oil to iron objects to prevent them from coming in contact with air (oxygen) and moisture, which are the two main requirements for rusting.
Explanation
The chemical reaction for rusting is: 4Fe + 3O₂ + xH₂O → 2Fe₂O₃·xH₂O (hydrated iron oxide — rust). Both oxygen and water are needed for this reaction. When we paint or oil the surface, we create a barrier that stops oxygen and moisture from reaching the iron. Without these two reactants, the rusting reaction cannot occur.
In-content Ad
AdSense placeholder
Quick Check
Chips manufacturers flush nitrogen gas in chips packets to
Show AnswerHide Answer
(B) Prevent rancidity by removing oxygen
Explanation
Nitrogen is an inert (unreactive) gas. When nitrogen is flushed into the chips packet, it replaces oxygen. Since fats in chips need oxygen to get oxidised (become rancid), removing oxygen prevents rancidity. This keeps the chips fresh for longer. Nitrogen does not react with the food, so it does not change the taste or quality of chips.
Quick Check
Which of the following is an antioxidant used to prevent rancidity in food?
Show AnswerHide Answer
(B) Butylated Hydroxyanisole (BHA)
Explanation
BHA (Butylated Hydroxyanisole) and BHT (Butylated Hydroxytoluene) are synthetic antioxidants added to food products to prevent rancidity. Antioxidants work by getting oxidised themselves, thereby protecting the fats and oils in food from oxidation. Natural antioxidants include Vitamin C (ascorbic acid) and Vitamin E (tocopherol). NaCl is a preservative but not an antioxidant.
Quick Check
The process of respiration is
Show AnswerHide Answer
(B) Exothermic
Explanation
Respiration: C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O + energy. Glucose is broken down in the presence of oxygen to release energy. This energy is used by our body to perform activities. Since energy (heat) is released, it is an exothermic reaction. Our body temperature remains at 37°C because of this continuous exothermic process.
In-content Ad
AdSense placeholder
Quick Check
Which of the following is an endothermic reaction?
Show AnswerHide Answer
(B) Decomposition of limestone
Explanation
CaCO₃ + heat → CaO + CO₂. Limestone (CaCO₃) absorbs a large amount of heat when heated to decompose into quicklime (CaO) and carbon dioxide. Since energy is absorbed (not released), it is endothermic. Options A, C, and D all release heat — they are exothermic reactions.
Quick Check
What are the 5 types of chemical reactions studied in this chapter?
Show AnswerHide Answer
- (1)Combination reaction — A + B → AB (two or more substances combine into one)
- (2)Decomposition reaction — AB → A + B (one substance breaks into two or more)
- (3)Displacement reaction — A + BC → AC + B (more reactive element displaces less reactive)
- (4)Double displacement reaction — AB + CD → AD + CB (exchange of ions between two compounds)
- (5)Oxidation-Reduction (Redox) reaction — simultaneous oxidation and reduction
Explanation
- Opposite of combination = decomposition
- Displacement involves reactivity of metals
- Double displacement involves ion exchange and often forms precipitate
- Redox involves transfer of oxygen, hydrogen, or electrons
Quick Check
What is the Law of Conservation of Mass and how does it apply to chemical equations?
Show AnswerHide Answer
Law of Conservation of Mass: Mass can neither be created nor destroyed in a chemical reaction. The total mass of reactants equals the total mass of products.
Application: In a balanced chemical equation, the number of atoms of each element is equal on both sides of the equation.
Explanation
Proposed by Antoine Lavoisier. This is why we balance chemical equations — to ensure the same number of atoms are present before and after the reaction. Example: In 2H₂ + O₂ → 2H₂O, we have 4H and 2O on both sides. If the equation is unbalanced, it violates this law.
In-content Ad
AdSense placeholder
Quick Check
Which of the following is an example of a combination reaction?
Show AnswerHide Answer
(C) N₂ + 3H₂ → 2NH₃
Explanation
N₂ + 3H₂ → 2NH₃ is the Haber process for making ammonia. Here, two substances (N₂ and H₂) combine to form one product (NH₃) — that is a combination reaction.
Quick Check
Assertion
Show AnswerHide Answer
(A) Both A and R are true, and R is the correct explanation of A
Explanation
Respiration: C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O + energy. Glucose is oxidised (combined with oxygen) to release energy. Since energy is released, it is exothermic. The reason correctly explains why respiration is exothermic — the oxidation of glucose releases heat energy. So both the assertion and reason are correct, and the reason is the correct explanation.
Quick Check
A student heats lead nitrate Pb(NO₃)₂ in a test tube. Write the balanced equation and identify the type of reaction.
Show AnswerHide Answer
2Pb(NO₃)₂(s) → 2PbO(s) + 4NO₂(g) + O₂(g)
Type: Decomposition reaction (thermal decomposition)
Explanation
- (1)It decomposes into lead oxide (PbO), nitrogen dioxide (NO₂) — a brown/reddish-brown gas, and oxygen gas (O₂)
- (2)Since one substance breaks into multiple products using heat, it is thermal decomposition
- (3)Observations: Yellow/orange fumes of NO₂ are seen, the white solid turns yellow (PbO)
- (4)A glowing splint brought near the mouth of test tube relights — confirming O₂ is released
In-content Ad
AdSense placeholder
Quick Check
Why does the colour of copper sulphate solution change when an iron nail is dipped in it?
Show AnswerHide Answer
When an iron nail is dipped in copper sulphate (CuSO₄) solution:
Fe + CuSO₄ → FeSO₄ + Cu
The blue colour of CuSO₄ fades and changes to light green (FeSO₄). Reddish-brown copper deposits on the iron nail.
Explanation
Iron (Fe) is more reactive than copper (Cu) in the reactivity series. So iron displaces copper from copper sulphate solution. As the blue CuSO₄ is consumed and green FeSO₄ is formed, the solution changes colour from blue to green. Copper metal deposits on the iron nail as a reddish-brown coating. This is a displacement reaction.
Quick Check
What happens when dilute hydrochloric acid is added to zinc granules? Write the equation and name the type of reaction.
Show AnswerHide Answer
Zn(s) + 2HCl(aq) → ZnCl₂(aq) + H₂(g)↑
Type: Displacement reaction
Observation: Brisk effervescence (bubbles) of hydrogen gas are produced. The zinc granules slowly dissolve.
Explanation
Zinc is more reactive than hydrogen. So zinc displaces hydrogen from hydrochloric acid. The H₂ gas produced causes bubbling (effervescence). We can test the gas by bringing a burning matchstick near the mouth of the test tube — H₂ burns with a pop sound. The zinc chloride (ZnCl₂) formed dissolves in water (remains in solution).
Quick Check
Which among the following is a double displacement reaction?
Show AnswerHide Answer
(C) Na₂SO₄ + BaCl₂ → BaSO₄↓ + 2NaCl
Explanation
In Na₂SO₄ + BaCl₂ → BaSO₄↓ + 2NaCl, the ions Na⁺ and Ba²⁺ exchange their partners — Na pairs with Cl and Ba pairs with SO₄. Two compounds exchange ions to form two new compounds. A white precipitate of BaSO₄ is also formed. This is a double displacement and also a precipitation reaction.
In-content Ad
AdSense placeholder
Quick Check
List the key balanced equations from this chapter.
Show AnswerHide Answer
- (1)Combination: 2Mg + O₂ → 2MgO
- (2)Combination (lime+water): CaO + H₂O → Ca(OH)₂
- (3)Decomposition (thermal): 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂
- (4)Decomposition (electrolytic): 2H₂O → 2H₂ + O₂
- (5)Decomposition (photolytic): 2AgCl → 2Ag + Cl₂
- (6)Displacement: Fe + CuSO₄ → FeSO₄ + Cu
- (7)Double displacement: Na₂SO₄ + BaCl₂ → BaSO₄↓ + 2NaCl
- (8)Redox: CuO + H₂ → Cu + H₂O
- (9)Rusting: 4Fe + 3O₂ + xH₂O → 2Fe₂O₃·xH₂O
- (10)Respiration: C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O + energy
Explanation
These 10 equations are frequently asked in exams. Learning them with the type of reaction will help answer most questions from this chapter. Always balance the equation and write physical states for full marks.
